A Race Against Time: One Man’s Fight to Prevent ALS Before It Strikes
Living Under a Dark Cloud: A Family Haunted by ALS
Jeff Vierstra has never shied away from adventure. The scientist and avid outdoorsman has fearlessly skied down the frozen slopes of icebergs off Chile’s coast and jumped from airplanes high above British Columbia. But there’s one thing that truly frightens this 41-year-old daredevil: his family history. Vierstra’s mother died from complications of Amyotrophic Lateral Sclerosis (ALS) when he was just two years old. The tragedy didn’t end there—all of his mother’s siblings also succumbed to this devastating neurodegenerative disease in their late thirties and early forties. Later, his two sisters, Erin and Leigh, would follow the same heartbreaking path. For Vierstra and his sisters, the threat of ALS wasn’t just a possibility—it was written in their DNA. All three siblings tested positive for a mutation in a gene called “FUS,” which is essential for normal cell function, particularly in nerve cells. This genetic time bomb made it highly likely they would develop the disease that had already claimed so many family members. Living with this knowledge created an invisible but constant presence in their lives. “Living with that sort of like cloud over you is like mentally and emotionally like really difficult,” Vierstra shared, describing the psychological burden of knowing what might lie ahead.
Understanding the Enemy: What ALS Does to the Body
ALS, also known as Lou Gehrig’s disease after the famous baseball player who brought public attention to the condition, is a cruel disease that progressively destroys motor neurons—the nerve cells responsible for controlling muscle movement. As these neurons degenerate, patients gradually lose their ability to perform even the most basic physical functions. What might start as weakness in a limb or slurred speech eventually progresses to complete loss of mobility. Over time, people with ALS lose the ability to walk, to use their hands, to speak clearly, and ultimately, to breathe on their own. The disease essentially traps a person’s mind inside a body that no longer responds to their commands. Currently, approximately 35,000 people in the United States are living with ALS, according to the Centers for Disease Control and Prevention. While most cases occur seemingly at random, about 10 to 15% of all ALS patients have a genetic form of the disease, research indicates. Of these genetic cases, roughly two-thirds are familial, meaning they run through multiple generations of a family, as explained by Dr. Neil Shneider, a Columbia University neurologist specializing in genetic forms of ALS. For families like Vierstra’s, the hereditary nature of their particular form of ALS created a devastating pattern across generations. As of now, there is no cure for ALS, but researchers around the globe are racing to develop new treatments that might slow its progression or, in some cases, prevent it altogether.
A Groundbreaking Experiment: Trying to Stop ALS Before It Starts
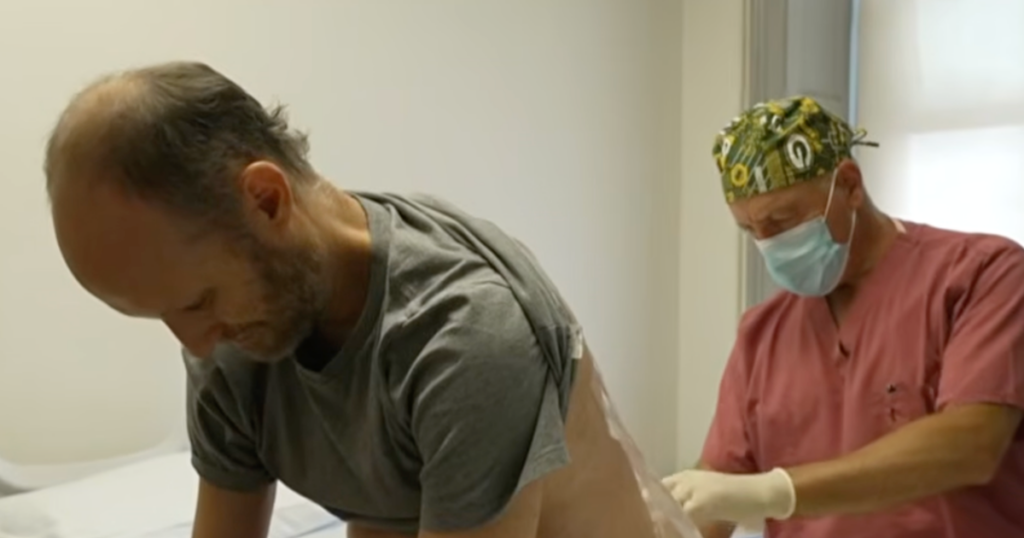
When both of Vierstra’s sisters began showing symptoms of ALS, they enrolled in a clinical trial at Columbia University’s Eleanor and Lou Gehrig ALS Center. The experimental treatment represented a cutting-edge approach: targeting the specific mutated FUS gene responsible for their inherited form of the disease. Dr. Shneider, who was leading this trial, also examined Vierstra as part of the research. What he discovered was both alarming and potentially life-saving. An electromyography test (EMG), which measures electrical signals in muscles, revealed abnormalities in Vierstra’s results. “It was a difficult moment,” Dr. Shneider recalled. “We thought this meant that this was an early sign of disease onset and that he was at risk for developing full blown disease.” However, this early detection also presented an unprecedented opportunity: What if they could intervene before ALS fully took hold? Dr. Shneider offered Vierstra the same experimental treatment his sisters were receiving, but with a crucial difference—Vierstra would be attempting to prevent the disease rather than slow its progression after symptoms appeared. This made Vierstra’s case the first known attempt to prevent ALS before it fully manifested. Determined to fight back against the genetic fate that had claimed so many family members, Vierstra didn’t hesitate. “I jumped at the opportunity to do that,” he said, ready to be proactive about his health rather than passively waiting for symptoms to appear.
The Treatment: A New Weapon Against a Genetic Enemy
For the past three years, Vierstra has undergone a unique medical protocol every few months. The treatment involves receiving infusions directly into his spine—a procedure known as intrathecal administration. These infusions contain specially designed molecules that target and disable the mutated FUS gene responsible for his family’s form of ALS. The science behind this approach represents a sophisticated understanding of genetics and molecular biology. Rather than trying to address ALS symptoms after they appear, this treatment aims to silence the faulty genetic instructions at their source, preventing the cascade of cellular dysfunction that leads to motor neuron death. The therapy is part of a broader category of treatments called gene therapy, which seeks to correct or compensate for genetic abnormalities causing disease. In Vierstra’s case, the treatment has been remarkably promising. After just one year of receiving these regular infusions, the mild abnormalities that had appeared on his muscle testing had normalized. This reversal suggested that the treatment might indeed be preventing the progression of disease that had seemed inevitable given his family history. While both of his sisters tragically developed complications from ALS and eventually died, Vierstra believes the treatment had a meaningful impact on them as well. “I felt the treatment extended their life,” he said, finding some comfort in the additional time the experimental therapy may have provided them. Their participation in the trial, even as the disease progressed, contributed valuable data that continues to inform research and may help others facing similar genetic risks.
Living Beyond the Expected: Hope for the Future
Today, at 41 years old, Jeff Vierstra has already outlived many members of his family who carried the same genetic mutation. He continues to work as a scientist, and his adventurous spirit remains undiminished—he still skis, hikes, and travels the world. But perhaps more significantly, he’s beginning to think about his future in ways that once seemed impossible. For someone who grew up expecting to die young from ALS, this shift in perspective represents a profound psychological transformation. “And maybe this actually is working for me and I can start thinking about the future, and I otherwise couldn’t have,” Vierstra reflected. The ability to make long-term plans, to imagine growing older, to live without the constant shadow of impending illness—these are gifts most people take for granted but mean everything to someone in Vierstra’s position. He describes the feeling as getting “another lease of life,” a second chance that seemed unimaginable when he first tested positive for the FUS mutation. Dr. Shneider shares Vierstra’s cautious optimism and believes the implications of this research extend far beyond one patient. “I think there’s real hope and opportunity to make this a liveable disease, one that isn’t fatal,” he said, articulating a vision of ALS as a manageable chronic condition rather than an inevitable death sentence. The success of Vierstra’s preventive treatment suggests that for genetic forms of ALS, early intervention before symptoms fully develop might offer the best chance of success.
The Bigger Picture: Hope for All ALS Patients
While Vierstra’s case involves a rare genetic form of ALS, the research being conducted at Columbia University has implications that could eventually benefit the broader ALS community. Dr. Shneider and his colleagues at the Eleanor and Lou Gehrig ALS Center hope that insights gained from studying and treating familial forms of the disease will eventually translate to therapies for non-familial cases as well. The center has launched “Silence ALS,” an initiative dedicated to developing individualized gene-based therapies for patients with other rare genetic forms of ALS. This personalized medicine approach recognizes that ALS isn’t a single disease but rather a spectrum of conditions with various genetic and environmental factors. By developing targeted treatments for specific genetic mutations, researchers are creating a toolkit that could eventually address many different forms of the disease. The work being done represents a fundamental shift in how we think about and treat neurodegenerative diseases. Rather than waiting until symptoms appear and trying to manage decline, the preventive approach demonstrated in Vierstra’s case suggests we might one day screen for genetic risk factors and intervene before irreversible damage occurs. For the thousands of people currently living with ALS and the many more at risk due to family history, this research offers something precious: hope. Jeff Vierstra’s journey from facing a seemingly inevitable fate to potentially preventing ALS altogether represents not just a personal victory but a beacon of possibility for all those touched by this devastating disease. His willingness to participate in experimental treatment, combined with the dedication of researchers like Dr. Shneider, may be paving the way toward a future where ALS is no longer the terminal diagnosis it has been for generations.